Nearly 200 attendees in 8 inspiring sessions addressing the different driving forces in the Belgian clinical research ecosystem!
The morning session
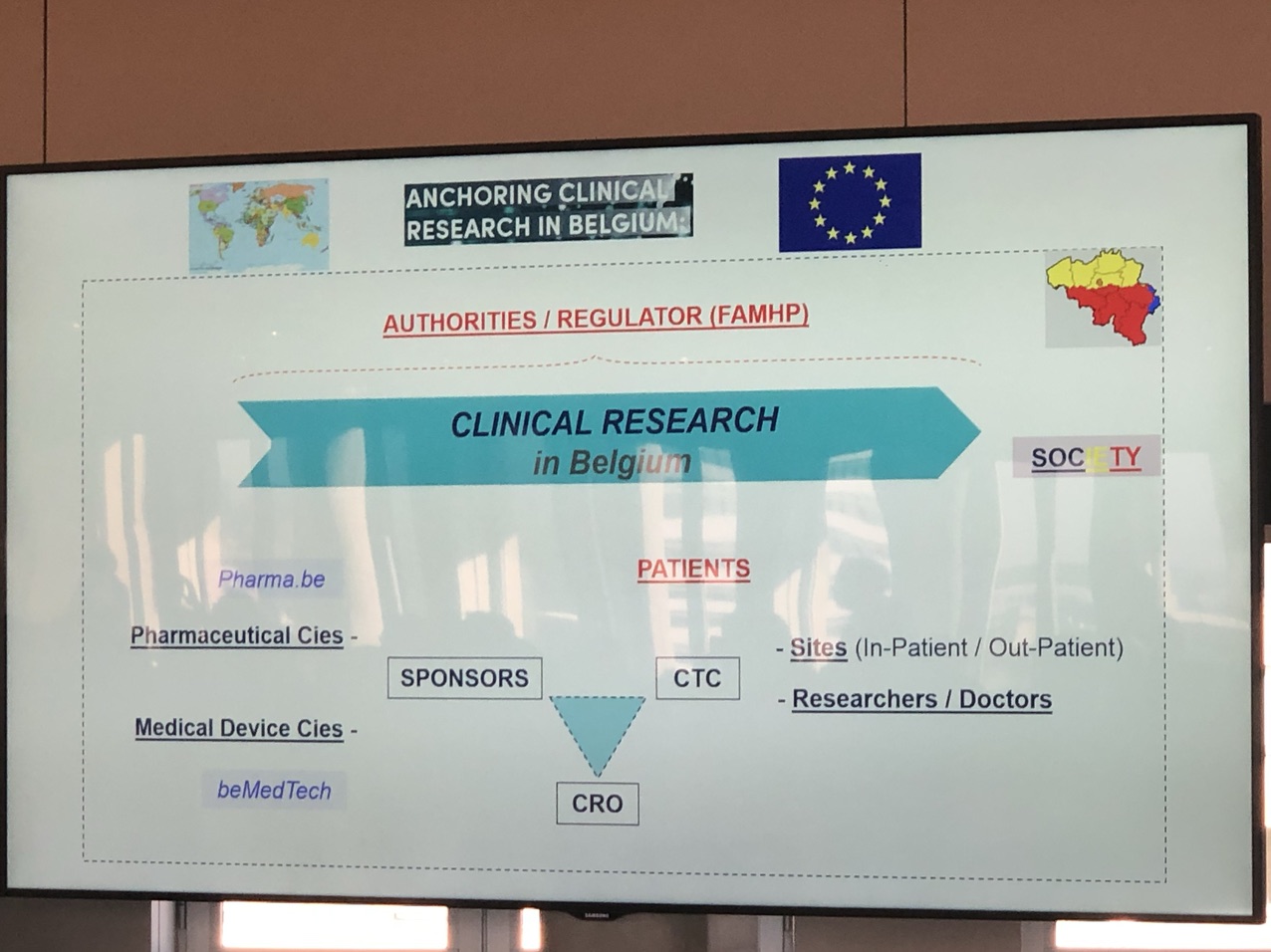
The morning session was an overview of the clinical and Medtech trials status. The challenges due to the new Clinical Trial Regulations (CTR) & the Medical Device Regulation (MDR) were in overview, too. Session contained also, the opportunities due to digitization and data analytics.
Catherine Rutten from pharma.be addressed the main reasons why companies choose Belgium for their clinical trials. She focused on the distinctive expertise, patient involvement and the adoption of digital technologies. All these rate Belgium consistently part of the top-3 countries in terms of number of clinical trials per 1 million capita.
In the next talk from Marnix Denys of beMedTech ,we were told that the same did not apply for the Medical Device trials with the new regulation of MDR coming up in March 2020. That makes Belgium not so competitive.
Greet Musch from FAMHP explained the new strategy of EMA. New stategy aims to simplify and integrate the new science and technology that happens in clinical trials into the new CTR.
Following was an informative presentation on how hospitals as Clinical Trial centers, deal with the overload of the administrative support that they provide to clinical research.
The closing presentation before lunch was an interactive discussion from Clinical4you and BECRO. In this presentation we got information on how to choose the right Clinical ResearchOrganization for trials. Αn interactive poll too place during the session that asked the audience on their thoughts and selection criteria, when choosing a CRO. Three important characteristics of the right CRO, that the audience agreed on, were
- a mutual understanding
- their treatment type expertise and
- their team experience in planning your project.
But as they said “The results you will get from a CRO are only as good as what you give them”.
The afternoon session
The afternoon session had a different format. There were three panels with a moderator introducing case study speakers following with a Q&A on specific topics.
AYMING Consulting moderated the first panel related to Clinical Trial Funding. It was presented how startups and University spinoffs can look for private, regional, national or European funding. Key take away was that H2020 funds are a good source of funding to develop further your product (which Innovation Sprint does), that there are tax benefits involved in R&D activities and a lot of regional funds before raising money from VC or an IPO and giving away too much equity.
IQVIA moderated the second panel. The panel was related to Patient enrolment and retention in clinical trials. With around half of trials missing, the enrollment targets it is most important to understand
- the needs of the patients
- the impact of the protocol design
- the use of digital technology, diversity & consent challenges
KEYRUS Group moderated the third panel on the hot topic of Digital transformation. The panel presented how lacking the market is compared to others. The panel highlighted how complex this topic is in the clinical setting and how we can learn from successful implementations on other industries. How to do data source extraction, data processing and analytics to present results with interactive dashboards.


Take away messages are that the complex regulatory framework is challenging and needs simplification. In addition, patient engagement is core and the building of trust.
No one can do it alone, so we need to work together, reduce the burden of the clinical trials, embrace technology and coevolve in this new challenging ecosystem.