Strategic collaboration with GEMELLI HOSPITAL and ASTRAZENECA IT.
Innovation Sprint announces early results from the strategic collaboration with AGOSTINO GEMELLI UNIVERSITY HOSPITAL and ASTRAZENECA ITALY related to the AZIMUTH model of care for heart failure.
Brussels, Belgium – Innovation Sprint Sprl, a digital biotech company and owner of Healthentia solution, announces encouraging early results from the strategic partnership with Fondazione Policlinico Universitario ‘Agostino Gemelli’ IRCCS (Rome, Italy) and AstraZeneca Italy, related to the co-developed AZIMUTH model of care, which aims to support heart failure patients. The first phase of the study has successfully validated the feasibility, patient acceptance, and perceived value of the app-based model of care, demonstrating increased user engagement in patients that leads into improved adherence to treatment. AZIMUTH model of care is delivered through Healthentia; a Software as a Service (SaaS) medical device via a mobile application for patients and a clinical dashboard for clinicians for Remote Patient Monitoring (RPM), Patient Support Programs (PSP) and Digital Therapeutics (DTx).
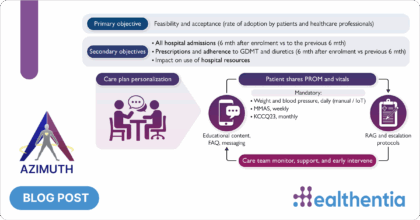
AZIMUTH model of care is a public-private multi-partners academia-industry collaboration, enabling digital health solutions to address the main unmet needs identified in the management of patients with heart failure and chronic ischemic disease. This innovative scalable model of personalised remote heart failure care is based on good clinical practice standards and international guidelines for heart failure management and has been designed taking into consideration both the optimization of in-hospital care delivery models as well as the often unmonitored out-of-hospital and home settings.
Based on the early results of the AZIMUTH study, the partnership has been awarded the “Best Scientific Collaboration Program of the Year” in the Life Science Excellence Award 2022 and now competing for the title of Excellence of the year in the same contest.
The Phase II of AZIMUTH will be launched in July 2022, extending the AZIMUTH model of care to at least five to seven different sites and involving more than 300 patients, aiming to demonstrate improvement in health-related endpoints, while performing a Health Technology Assessment (HTA). The Fondazione Policlinico A. Gemelli IRCCS is the Lead investigator of the study.
Heart failure is the leading cause of death and hospitalisation in patients aged > 65 years and a major and growing medical and economic burden, with high prevalence and incidence rates worldwide. The recent pandemic crisis has made even more evident, some critical aspects in the management of complex chronic diseases, such as heart failure, and these could be exacerbated by the increased demand for care in the future. Therefore, a significant effort must be made to align services with patients’ care needs by identifying a shared model that can exploit the most advanced technologies to enhance disease deterioration, provide adequate integration between hospital and territorial services, increase appropriateness, and reduce waiting times for specialist services.
Dr. Domenico D’Amario, senior cardiologist at the Gemelli IRCCS Polyclinic Foundation quoted “The care pathway for patients with heart failure includes several steps, from acute phase treatments to chronic phase patient follow-up. This requires a close dialogue between hospital cardiologists and community medicine, especially once the patient returns home, after being discharged from a third-level hospital. But at the moment this complex path is very fragmented.”
Konstantina Kostopoulou, product owner of Healthentia, quoted “The AZIMUTH model of care used agile design and development processes involving multiple stakeholders and following best practices to increase User eXperience(UX) through intuitive User Interfaces (UI) both for the patient’s app but also for the clinician’s portal. The results from Phase I are encouraging with regards to patients’ engagement and acceptance and this is a great proof to keep going and scaling the AZIMUTH model of care across more patients and hospitals.”
Sofoklis Kyriazakos, CEO of Innovation Sprint, quoted “Digital healthcare has experienced high growth since the start of the pandemic, while the disruption in the MedTech industry comes to a good extend from the use of Software as Medical Devices (SaMD) for Patient Support Programs (PSP) and Digital Therapeutics (DTx). We invest our efforts to make Healthentia a pioneering solution based on the best of breed of Information Technologies (IT), IoT (Internet of Things) devices, AI/ML processing and Virtual Coaching. Our purpose is to offer digital clinical pathways through Healthentia, giving access to any person at any place to clinical care services for improved health and wellbeing.”
About Innovation Sprint
Innovation Sprint is a digital biotech company that brings innovative technologies in Life Sciences and MedTech to improve clinical research trials and improve patients’ health through its product Healthentia. Healthentia is a digital platform that enables digital therapeutics by offering a smartphone application for patients and a portal for healthcare professionals to securely access smart services and insights. Healthentia provides insights and predictions of patients’ health that feed an advanced virtual coaching system with personalized information. Thus, digital clinical endpoints are synthesized, and contribution is made to the design of DTx content.
Today, Healthentia is used in several studies with Pharma and large hospitals in the investigator-driven domain, to file clinical evidence in the regulatory setting and we also derive health economics analytics for policy makers and healthcare payers & providers about the improvement of health-related endpoints in several therapeutic areas with particular focus on heart failure, other non-communicable chronic diseases, and cancer. Healthentia is compliant with the medical device manufacturing policies and procedures, as these are defined by ISO 13485 in Europe and CFR 820 from the FDA.
Innovation Sprint is based in Brussels (BE) since its incorporation in 2016 and has two labs, one in Athens (GR) and one in Enschede (NL).
More information
Innovation Sprint – https://innovationsprint.eu
Healthentia – https://healthentia.com